Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 U/
U/ U ratio for a trace amount of uranium: Search for an extinct radionuclide
U ratio for a trace amount of uranium: Search for an extinct radionuclide 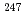 Cm in the early solar system
Cm in the early solar systemChai, J.; Miyamoto, Yutaka; Kokubu, Yoko; Magara, Masaaki; Sakurai, Satoshi; Usuda, Shigekazu; Oura, Yasuji*; Ebihara, Mitsuru*
Journal of Radioanalytical and Nuclear Chemistry, 272(2), p.397 - 401, 2007/05
Times Cited Count:2 Percentile:18.72(Chemistry, Analytical)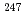 Cm is an important nuclide because it has the next-shortest half-life (1.6
Cm is an important nuclide because it has the next-shortest half-life (1.6 10
10 yr) from
yr) from  Pu among the transuranic nuclides. The disintegration of
Pu among the transuranic nuclides. The disintegration of 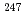 Cm to
Cm to  U by three
U by three  decays and two
decays and two  decays and the chemical fractionation between Cm and U during the formation of solid materials in early solar system lead to changes in the ratios of
decays and the chemical fractionation between Cm and U during the formation of solid materials in early solar system lead to changes in the ratios of  U to
U to  U. This variation may provide strict constraints on the time interval between the last r-process nucleosynthetic event and the formation of the solar system. It is important to develop a practical analysis protocol to determine the isotope ratio of trace uranium in geochemical and environment samples. As uranium is the least abundant element in our solar system, analytical techniques with high sensitivity and precision are required. In this study, a double focusing inductively coupled plasma mass spectory was applied to measure isotopic ratio of uranium. By one set data obtained from the EDTA phase of the Jilin meteorite sample, the upper limit of
U. This variation may provide strict constraints on the time interval between the last r-process nucleosynthetic event and the formation of the solar system. It is important to develop a practical analysis protocol to determine the isotope ratio of trace uranium in geochemical and environment samples. As uranium is the least abundant element in our solar system, analytical techniques with high sensitivity and precision are required. In this study, a double focusing inductively coupled plasma mass spectory was applied to measure isotopic ratio of uranium. By one set data obtained from the EDTA phase of the Jilin meteorite sample, the upper limit of 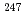 Cm in the early solar system was calculated and
Cm in the early solar system was calculated and 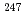 Cm/
Cm/ U
U  0.003 was obtained.
0.003 was obtained.